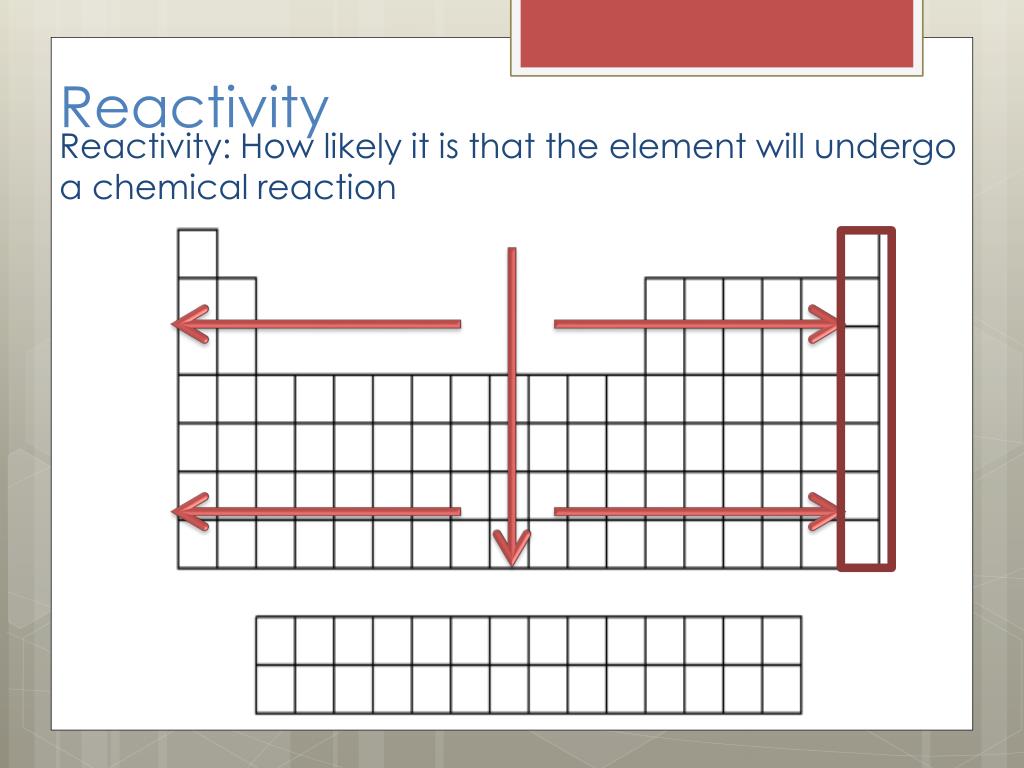 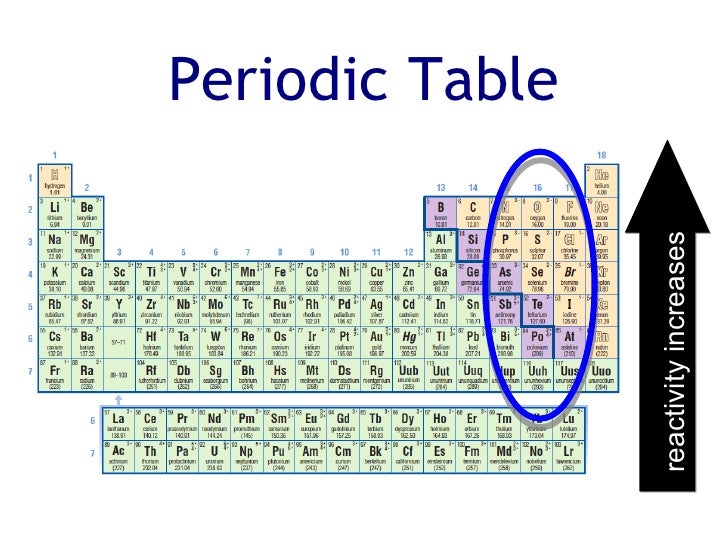 
Note that there is no clear division between metallic and non-metallic character. contains non-metal close non-metal Element that is a poor. Element Titanium (Ti), Group 4, Atomic Number 22, d-block, Mass 47.867. Fluorine gas reacts explosively with many other elements and compounds, and is considered to be one of the most dangerous known substances. Group 0 close group 0 The vertical column of non-metal elements on the far right of the periodic table, also called the noble gases. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. It is not found in nature as a free element. Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The most reactive nonmetals reside in the upper right portion of the periodic table. Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. In addition, the atomic radius increases going down a group, placing the outer electrons further away from the nucleus and making that electron less attracted by the nucleus. Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. Neon gas filled the first illuminated science, which were produced almost a Century ago and since then, it has infiltrated language and culture. Its the gas that can give you your name or any word you like, in fact, in light. The metallic character increases as you go down a group. This could be the most captivating element of the periodic table. Reactivity of metals is based on processes such as the formation of halide compounds with halogens, and how easily the element displaces hydrogen from dilute acids. (Credit: User:Mirek2/Wikimedia Commons Source: Commons Wikimedia, Periodic Trends(opens in new window) License: Public Domain) This is what causes these elements to react in the same ways as the other members of the family. All of the 1A elements have one valence electron. A group is a vertical column of the periodic table. Reacts on heating to form a black solid.\): Trends in behaviors of elements. Remember that Mendeleev arranged the periodic table so that elements with the most similar properties were placed in the same group. Because each orbital can have a maximum of 2 electrons, there are 2 columns in the s. The electron configurations of the elements are in Figure 6.9.2. Reacts as a powder on very strong heating. 1: The Periodic Table, Showing How the Elements Are Grouped According to the Kind of Subshell ( s, p, d, f) Being Filled with Electrons in the Valence Shell of Each Element. Reacts readily when heated as iron filings. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. 
Yellow solid forms which changes to white on cooling. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. Reacts steadily when heated forming a yellow solid which changes to white on cooling. But, at the far right are the noble gasses, and they are almost completely inert. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Magnesium (Mg), Group 2, Atomic Number 12, s-block, Mass 24.305. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Each element going from left to right tends to be more reactive. History textPodcasts The Naked Scientists. Reacts readily with strong heating as a powder. Reactivity: The reactivity of the elements increases going from left to right on the periodic table. Slowly forms a surface oxide at room temperature Yellow/orange flame and white solid formed. A great Starter for ten activity to engage your students with trends in the periodic table by focusing on group 2, group 3 and group 7. a microscale practical into the extraction of copper, where students can engage with the experiment and build on their practical chemistry skills. Tarnishes when freshly cut at room temperature The extraction of copper: a microscale version.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
